KAWANISHI-CITY, Japan, Sept. 17, 2020 /PRNewswire/ — Rechargeable batteries are a very important device because they can store electricity produced by sustainable energy sources such as wind power and solar cells. So far, lithium ion batteries are the typical rechargeable battery in modern society. Therefore, the challenge to increase the capacity of lithium ion batteries is an extremely important task.
Green Science Alliance is the company that focuses in researching cutting edge technology in the field of energy and environmental green science. One of their businesses is developing next generation rechargeable batteries. They synthesize electrode, electrolyte material themselves. In addition, they make coating ink, test batteries, measure their electrochemical properties, and analyze technical detail. All procedures can be done within their company, which could be an advantage compared to other competitors.
For cathode material for lithium ion batteries, necessary properties are high voltage, high capacity, good cycle characteristics with high durability, high charge discharge rate at high current density. In addition, chemical and thermal stability is also important. Not to mention, when one considers about commercialization, low cost, material synthesis outcome stability and efficiency are also critical issue.
Researchers have been challenged to make various type of cathode material for lithium ion batteries. Transition metal oxide with layered rock salt structure, spinel type structure and olivine type structure etc. have been mainly attempted. Some of them are commercialized in the market and those are LiCoO2, LiNi0.33Mn0.33Co0.33O2, LiNi0.5Mn0.2Co0.3O2, LiNi0.6Mn0.2Co0.2O2, LiNi0.8Mn0.1Co0.1O2, LiNi0.8Co0.15Al0.05O2 as layered rock salt structure, and LiFePO4 have been also commercialized as olivine cathode. These cathode materials indicate average voltage as 3 – 4 V and theoretical capacity as 170 – 280 mAhg-1. For practically used battery, they often show 100 – 200 mAhg-1 as capacity. Nowadays, people demand for higher capacity and voltage in order to create stronger lithium ion battery. As high voltage cathode, LiNi0.5Mn1.5O4 as spinel structure and Li2CoPO4F as olivine structured cathode materials, have been under intensive research. These materials present as high as 4.5V as average voltage and that is approximately 1 V higher than normal cathode.
Recently, lithium rich cathode as layered rock salt structure type electrode such as Li2Mn1/2Ti1/2O2F and Li1.2Ti0.4Mn0.4O2 have been the focus as they exhibit high capacity. Molar ratio of lithium compared to transition metal in the lithium rich cathode material, is larger than 1. Its theoretical capacity is 380 – 460mAh/g. Since they have about 100 mAh/g larger cell capacity than conventional cathode material, lithium rich cathode has been gathering much attention in the world.
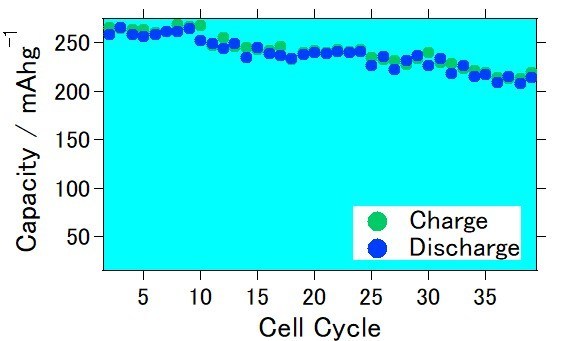
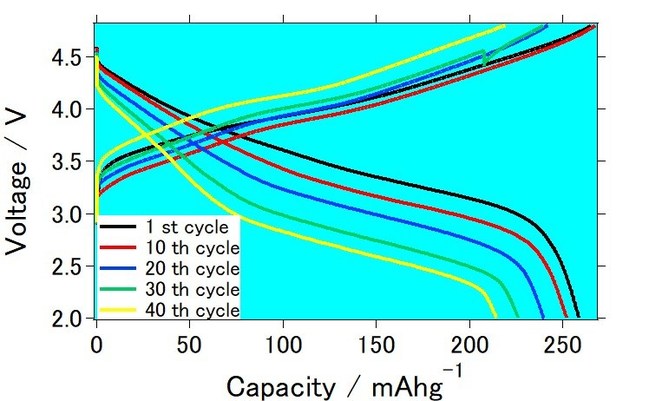
This time, Mr. Katsuhiko Konishi and Dr. Ryohei Mori at Green Science Alliance have developed lithium rich cathode with particular chemical composition. They had also prepared printable ink with their lithium rich cathode. Testing batteries was prepared with their lithium rich cathode, lithium metal as counter electrode, and carbonate-based electrolyte was used as an electrolyte. As a result, cell exhibited approximately 265 mAh/g as initial stage and became about 215 mAh/g after 40 charge-discharge cycles. This electrochemical measurement was performed with 0.1 C of charge discharge current at room temperature. They had also confirmed that when graphite was used as an anode, capacity became approximately 240 mAh/g as initial cycle. Since they observed high capacity with actual full cell, they are planning to manufacture actual lithium ion batteries with their lithium rich cathode.
They will keep testing to increase capacity and to obtain good charge/discharge cycles in order to create lithium rich cathode-based lithium ion batteries with high energy.
Contact:
Ryohei Mori
+81727598501
247365@email4pr.com
SOURCE Green Science Alliance Co., Ltd.

